How is Alkaline (Reduced) & Acidic (Oxidized) Ionized water produced?
A water ionizer electrolyzes water with direct current voltage (DCV). A special attachment re-directs tap water out of the faucet through a plastic hose into the ionizer unit. Inside, the water is first filtered through activated charcoal.
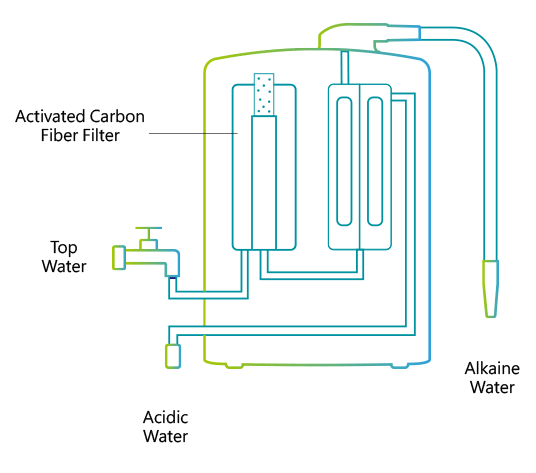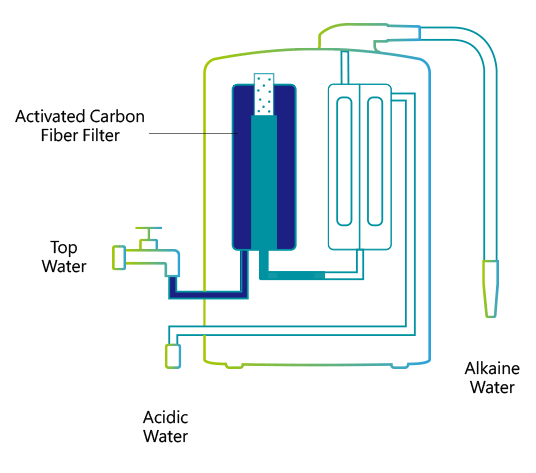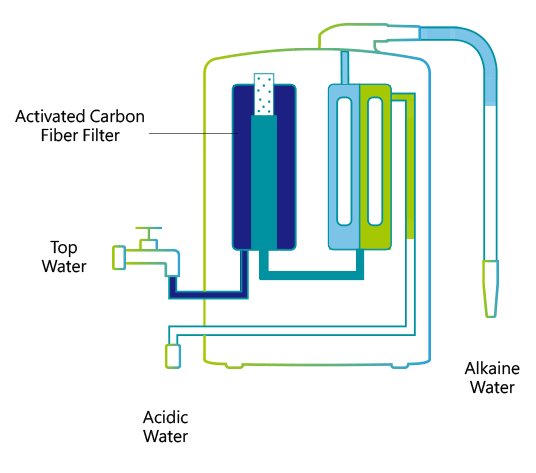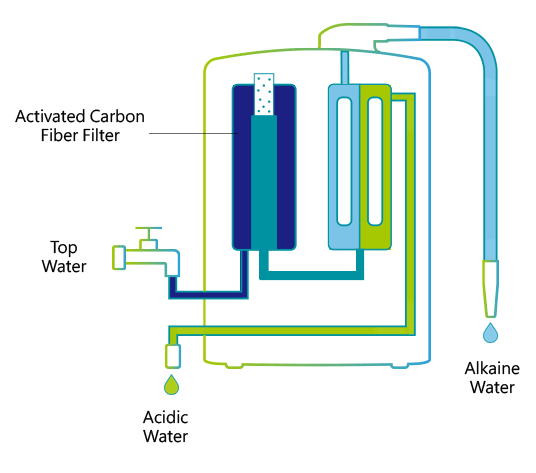
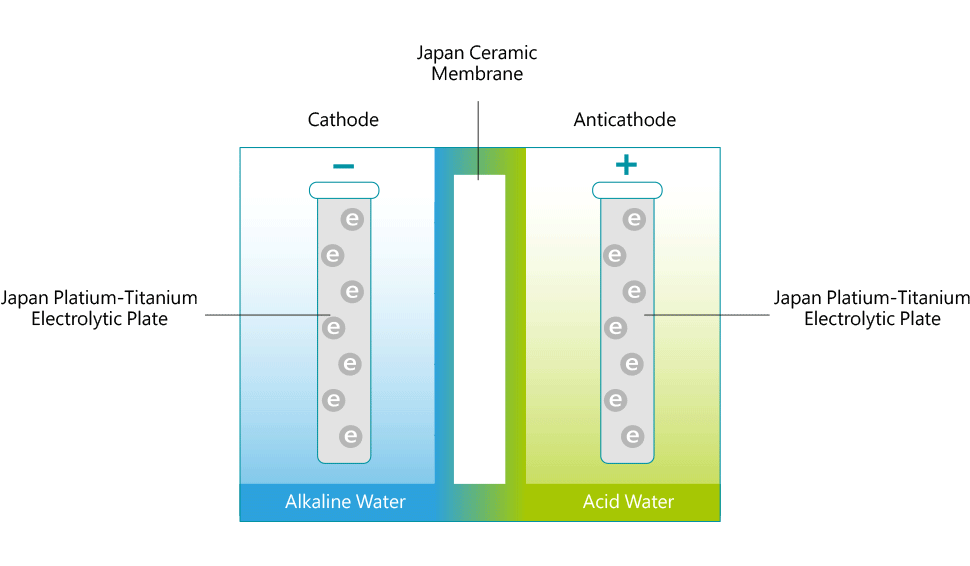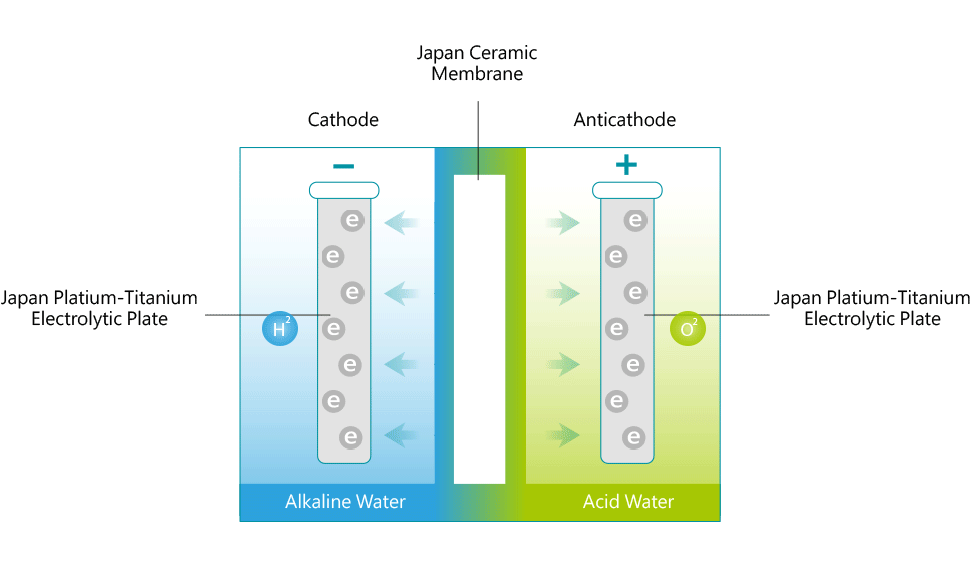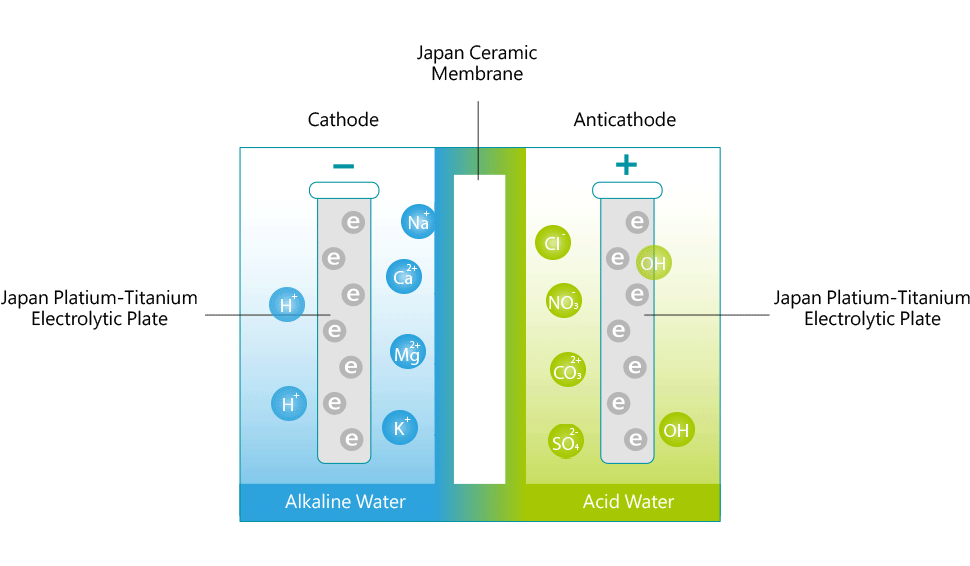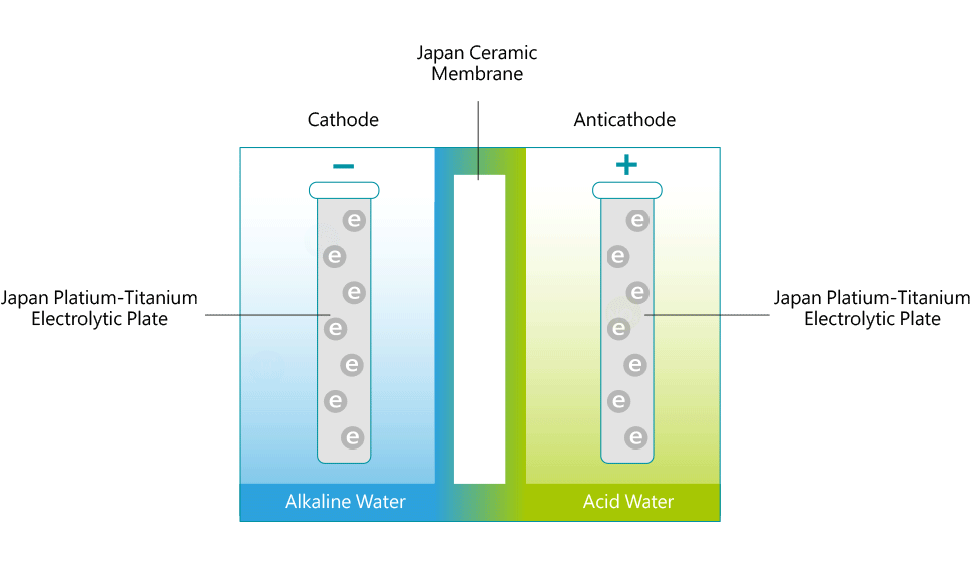
Each ionizer unit has a specially engineered electrolysis chamber equipped with platinum-coated titanium electrolysis plates. These plates are connected to a negatively(–) charged electrical terminal and a positively(+) charged electrical terminal. The filtered water flows into the chamber and is directed through a ceramic membrane that is positioned between the charged electrolysis plates. As the water flows, an electrical charge is passed through the terminals that electrifies the plates. As it flows, the water is conditioned by the electrical charge from the plates. The conditioned or 'Ionized' water then exits through two ports; (an alkaline port and an acidic port). Cations are attracted to the negatively charged plate to produce cathodic water (reduced water). Anions, negatively charged ions, are attracted to the positively charged plate to produce anodic water (oxidized water).
The (-) cathode attracts minerals such as sodium, magnesium, potassium, calcium and others and the water surrounding the terminal becomes alkaline with an increase in hydrogen ions (H+). The anti-cathode terminal attracts ions such as oxygen, sulfuric acid, sulfur and the water surrounding the terminal becomes acidic with an increase in hydroxide ions (OH-). The reduced water produced through electrolysis not only contains an excess amount of electrons (e-), but the cluster size of the H2O molecules seems to be reduced in size from about 10 to 13 molecules per cluster to 5 to 6 molecules per cluster. The water discharged from the alkaline port has a higher pH value and contains minerals, like Calcium and Magnesium, that are in a form that is more suitable for the body to assimilate
Calcium, Potassium, Magnesium and other minerals can normally be found in drinking water. But these minerals in are complex, inorganic and very stable. These are not easily assimilated by the body. However, minerals in ionized water are separated into ions with (-)ORP. These ions unite with the acidic residues with positive ions. They act as buffering agents that are easily assimilated and eliminated by our bodies.



Water Purification

Electroysis


Chlarine(CI-)
Sulfuric Acld(SO42-)
Carbonic Acid(Ca+)
Nitrlc Acid(NO2-)


Potassium(K+)
Solidum(Na+)
Calcium(Ca+)
Magnesium Ions(Ma2+)